How to control of annual bluegrass weevils with beneficial nematodes- Bugsforgrowers.com
This weevil is one of the most economically important pests of turf and it has got its name as Annual bluegrass weevil (Listronotus maculicolis) because it prefers to feed more on annual bluegrass (Poa annua) than any other grass species. Although annual bluegrass weevil develops through four different developmental stages including eggs, larvae, pupae and adults, only larva is the most damaging stage to annual bluegrass.
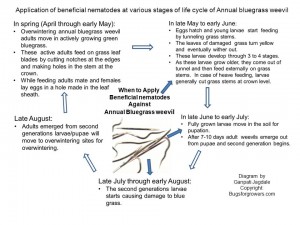
Annual bluegrass weevil overwinters as adults in the litter under trees and shrubs around and in the areas where annual bluegrass is grown. Overwintering adults become active early in the spring and move from their overwintering sites into actively growing annual bluegrass where they start feeding on the grass leaves and mate. Then females start laying egg by making holes under the bluegrass leaf sheath. Eggs hatch within a week that is early in the May and young larvae immediately begin feeding by tunneling into grass stems. Major symptoms of bluegrass weevil damage include yellowing of grass leaves and subsequently dying of grass. The larvae of bluegrass weevils develop through four stages. When larvae become large in size, they come out of the tunnels and start feeding externally on stems of grass and like cutworms; they sometime cut stems from roots causing severe turf loss.
To avoid complete loss of turf, it is recommended to control population of annual bluegrass weevil below its economic threshold level. Since first generation larvae of annual bluegrass weevil are very aggressive in causing damage to annual bluegrass in late May or in early June, it is recommended to employ beneficial/entomopathogenic nematodes to target larval population of annual bluegrass weevils.
When applied in late May or early June, three beneficial nematodes also called as entomopathogenic nematodes including Steinernema carpocapsae, Steinernema feltiae and Heterorhabditis bacteriophora proved to be effective in managing populations of annual bluegrass weevils. It has been demonstrated that any of these three beneficial nematodes can control over 65% population of annual bluegrass weevils when applied at the rate of 1 billion nematodes in the late May or early June.
Three beneficial entomopathogenic nematodes
- Steinernema carpocapsae: Steinernema carpocapsae nematodes use ambush type of foraging strategy to find annual bluegrass larvae and kill them.
- Steinernema feltiae: Steinernema feltiae nematode is a cold tolerant and uses intermediate type of foraging strategy (in between ambush and cruise types) to find and kill annual bluegrass weevil.
- Heterorhabditis bacteriophora: Heterorhabditis bacteriophora nematodes cruising type of foraging strategy to find their insect hosts including annual bluegrass weevil larvae.
Following diagram (Fig. 1) shows that the beneficial nematodes can be applied to target annual bluegrass larvae from 2-3 different generations during growing season of the annual bluegrass.
Research Papers
- McGraw, B.A. and Koppenhofer, A.M.2008. Evaluation of two endemic and five commercial entomopathogenic nematode species (Rhabditida: Heterorhabditidae and Steinernematidae) against annual bluegrass weevil (Coleoptera: Curculionidae) larvae and adults. Biological Control 46: 467-475.
- McGraw, B.A. and Koppenhofer, A.M.2009. Population dynamics and interactions between endemic entomopathogenic nematodes and annual bluegrass weevil populations in golf course turfgrass. Applied Soil Ecology 41: 77-89.
- McGraw, B.A., Vittumb, P.J. Cowlesc, R.S.and Koppenhofer, A.M. 2010. Field evaluation of entomopathogenic nematodes for the biological control of the annual bluegrass weevil, Listronotus maculicollis (Coleoptera: Curculionidae), in golf course turfgrass. Journal Biocontrol Science and Technology. 20: 149 – 163.
